Robert Langer
Description: Robert Langer is an Institute Professor at MIT, one of only 14 to hold this title, and a pioneering figure in biotechnology. His work in bioengineering has led to revolutionary new platforms for drug delivery, tissue engineering, and nanotechnology that form the basis of countless medical treatments and over 40 companies. In this episode, we discuss the design philosophy behind inventing technologies that solve previously impossible problems, like creating a pill that can inject large molecules from inside the stomach. Professor Langer also explains the science of a single-shot, self-boosting vaccine, his approach to bridging the gap between academia and industry, and his belief in fostering a creative, free-thinking environment to tackle the world's biggest health challenges.
Websites:
Publications:
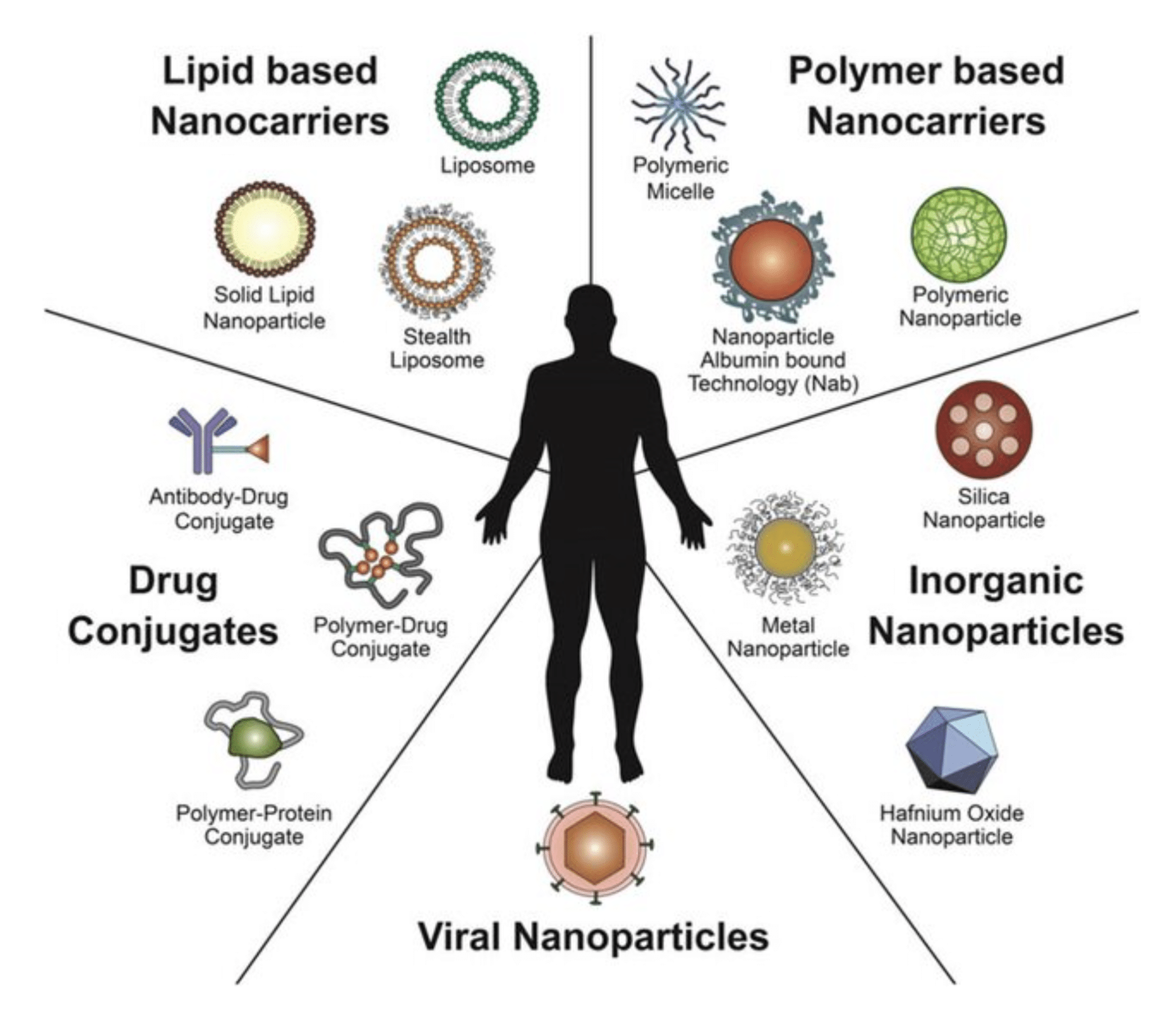
Nanocarriers as an emerging platform for cancer therapy
Mentions:
Other Podcasts:
Lex Fridman Podcast #105: The Edison of Medicine (2021)
Other:
Types of Nanomedicine
Nanomaterials: Applications in Biomedicine and Biotechnology
Show Notes:
[0:07] Introduction to Robert Langer
[0:46] Engineering Challenges in Drug Delivery
[2:08] Innovations in Oral Drug Delivery
[7:28] Leadership Philosophy in Academia
[10:41] The Creative Process in Research
[12:17] Collaboration and Team Dynamics
[17:46] Bridging Academia and Business
[19:12] Patient-Centric Research Approaches
[24:07] Educating the Public on New Technologies
[26:31] Future Challenges and Innovations
[28:05] Self-Boosting Vaccines Explained
[29:49] Starting New Ventures in Tech
[32:30] Navigating FDA Regulations
[35:07] Integrating Regulators into Research
[36:02] Advice for Aspiring Students
[36:54] Closing Thoughts and Advice
Unedited AI Generated Transcript:
Brent Valentine:
[0:00] Welcome back to Discovering Academia. We are two college students who travel
Brent Valentine:
[0:03] the world talking with academics about their research, passions, and current events. Today we talk with Robert Langer, one of nine institute professors at MIT and a world-renowned pioneer in drug delivery and tissue engineering.
Keller Kramer:
[0:16] Langer's work has led to revolutionary new platforms for drug delivery, tissue engineering, and nanotechnology that form the basis of countless medical treatments in over 40 companies. In this episode, we explore the engineering challenges of oral drug delivery, including a pill that can inject large molecules from inside the stomach, as well as the idea of self-boosting vaccines. We also discuss Professor Langer's leadership philosophy at his lab, how he bridges academic and industry partnerships, and his advice for young
Keller Kramer:
[0:42] scientists searching for meaningful, world-changing problems to work on. We hope you enjoy it.
Brent Valentine:
[0:47] Welcome, Professor Robert Langer. Thank you for coming on today.
Robert Langer:
[0:50] Thank you for having me.
Keller Kramer:
[0:52] You've tackled a vast range of health problems throughout your career. Is there an underlying philosophy that you use in your lab to decide what a health problem is right for the type of pioneering engineering you guys conduct in your lab?
Robert Langer:
[1:05] Well, I guess, I mean, it depends what it is. I mean, I'd like to think that I'd try to help on, first, I want it to be an important problem. And secondly, I mean, I just would have to look at it and try to get a feeling if it's going to involve, say, a lot of molecular biology or something that's not my skill set. I probably would be less inclined to do it. So it should be something that the kind of things like our lab, which is a bioengineering lab, might be good at.
Brent Valentine:
[1:38] Yeah would you if the problem is big enough would you still try to tackle it but then bring in some other people because your lab works with a lot of people across yeah
Robert Langer:
[1:48] Yeah absolutely i mean it uh you know we certainly do that it really i look at it in a case-by-case basis you know to see if i feel that it's something that we can make an impact in
Brent Valentine:
[2:00] Yeah and then when you're thinking about delivering a really difficult payload,
Brent Valentine:
[2:06] whether it's like a large macromolecule or something like that. What are some of the fundamental biophysical or material science hurdles that you prioritize solving first?
Robert Langer:
[2:16] Yeah, well, I mean, I guess the first thing is, and again, I break things up into a couple of things. One, one, Is it a whole new technology we're trying to invent, or is it adapting a technology that's already out there to the problem that you're asking? A lot of times what our lab does is we try to invent whole new technologies that don't exist. Examples might be really the first nanoparticles that could deliver genes or the first pills you could swallow that could deliver large molecules. I mean, but they're what I'd call platform technologies. Now, sometimes in the lab or even in a company, then you get more specific about what it is that you want to do. And then what you have is you have what I call a target product profile, which might mean, well, I want it to be delivered for a week at this level. It should be constant, or maybe it shouldn't be constant, but I have this profile that you want, and then you try to adapt an existing system that we've already maybe invented to that problem. So, I've been involved in doing both those things.
Keller Kramer:
[3:36] And when you're looking to develop drugs that kind of cross different modalities. Like you mentioned, orally ingested for a large molecule. What are you prioritizing first on that? Are you focusing on the aspects that need to be constant for it to be orally ingested? Or are you thinking more about the after effects of the delivery itself?
Robert Langer:
[3:56] Well, so let's take that example, orally ingested large molecule. Most people would say that's not possible. People have tried to do that for a long time.
Robert Langer:
[4:06] And so, you know, and one of the reasons when, you know, so you'd look back at what everybody's done and say, including ourselves, I'll pick this example because we've been doing it. And then what you realize is that almost every time it's been a large molecule, people get very, very little in. Why do you get very little in? Well, because you swallow it, it could be degraded by stomach acid. There are ways to deal with that by certain types of coatings. But even if you do that, then it will get degraded by enzymes. That's a much harder problem. And then if you somehow solve that, then how do you get it absorbed across the GI tract?
Robert Langer:
[4:44] So that was a big problem and still is a big problem. But a number of years ago, we started having the idea of creating a pill you could swallow that could make an injection in the stomach, just like now you make an injection in the arm. And we came up with a way to do it. We published it. I mean, I could go through the science, but it's basically involved a lot of different components in terms of thinking, I mean, thinking through a number of different engineering components. In the end, the system doesn't look very complicated. It's a little tiny thing. But at any rate, it involved this. And then, you know, we published it in Science and Nature and one of the cell journals. We used it to deliver different large molecules like insulin or messenger RNA or antibodies. You know, now it's actually started to be tested in patients even. That's amazing. Test is not used.
Brent Valentine:
[5:39] Yeah, yeah. And then did that technique involve combining a few different delivery modalities? Because I was really interested in the idea of like oral ingested and then targeting to cross like something like the blood-brain barrier.
Robert Langer:
[5:55] Yeah. Well, that did involve different technologies. I mean, just basically what we did is we made a little pill. I might even be able to find it. but it's got a membrane in that allows water to permeate. There's a little hole in it out of which the drug can get ejected. So behind that little hole, we have a needle, but the needle, I mean, there's different designs, but one design is you have a needle. Needle is actually the drug, maybe with some excipients. Behind that, you have a spring that would shoot it out. But you don't want it shut out right in the middle, right? That's your mouth. You want it maybe an hour later. So we have that spring embedded in a glass. But that glass is made of sugar. So what happens is you swallow it.
Robert Langer:
[6:53] Water comes in. And actually, an hour later, it'll just break that glass by osmosis. Then the spring, which is bigger than the hole, can't get out of the pill. But the needle, which is smaller than that hole, comes right out and is injected in the stomach. Stomach heals. It doesn't seem to hurt. Now, the one other issue that we had to come up with is how do you make sure that that hole is always going to be facing the stomach wall?
Robert Langer:
[7:22] So what we did is we modeled the system as a leopard tortoise. You might also think of a we have a wobble toy but the idea is you swallow it tumbles automatically and always because of the weight distribution it'll fall against stomach wall um and um and it shoots it out and uh so that that's how i could probably even find if i take a minute or two i might be able to find it i didn't know i was going to be going over this no
Brent Valentine:
[7:52] Yeah was there a priority in your as this design philosophy of which problem you need to tackle first, or is it you know you have the list of problems and then you just go one by one trying to check them off?
Robert Langer:
[8:07] Well, first we had just this idea in general. This was done largely by Gio Traverso, who was a fellow with me that now is a professor at MIT. He's a gastroenterologist, so he's got a great background for some of these things. But at any rate, Initially, we just wanted to prove a principle, but the molecule we used was insulin for a couple reasons. One, that would, I think, make a big difference someday if it could be done for little kids. Two, Novo Nordisk got to help fund it and collaborated with us, and that's one of the molecules that were of interest. But really, it could be anything.
Keller Kramer:
[8:49] And a big part of your lab is you know creating new biomaterials that can help with the kind of initial structure of the delivery for these drugs when you're thinking about the chemical, components needed for these polymers how do you balance that between the biological function that has to go into them well.
Robert Langer:
[9:10] A lot depends on what we're trying to do you know i mean if you're going to use something on a person, of course, the most important thing is that it be safe. For some applications, it's very important that you want the materials to be degradable too. For others, it'd be less important. For some applications, it may be important that it be very strong. For others, it might be less important. For some applications, you might I want to have it be able to permeate into a cell. And for others, you wouldn't. So you have to look at what kind of problem we're trying to go after.
Brent Valentine:
[9:49] Yeah. And then when you're thinking about the safety for these polymers or kind of non-biological things that will be entering someone's body, how much goes into thinking about where they go after the drugs administered?
Robert Langer:
[10:05] Well, I think you do. You have to think about everything. I mean, so sure, that's important.
Keller Kramer:
[10:13] And then looking at your lab's, I guess, impact over time, you know, you've clearly had sustained impact across, you know, a variety of fields, you know, across generations. How do you think about, I guess, the overarching philosophy that goes into the leadership of the lab that's allowed you to have this type of impact?
Robert Langer:
[10:33] Yeah, well, I guess the way I think about it is I want people to think about big problems that could make a big difference in the world.
Robert Langer:
[10:41] I like i want you know and i think that's what they want to do too um you know i i run i'd say a fairly loose operation you know where people have a lot of freedom um but people work really hard we have a lot of times we have teams of people working together on a problem i mean and i do have some people that help me on different things some people call them subgroups because the lab's pretty big but but those are some of the things that we do yeah
Brent Valentine:
[11:12] And then oh so
Keller Kramer:
[11:14] Let's see yeah how how does that creative process work you know because i think yeah i think freedom obviously must be an important tenet to allow that to foster but at what level i guess are people allowed to to bring ideas up or how does that that work on people starting with.
Robert Langer:
[11:30] You know well well usually we start on something that Maybe that was a problem we're trying to solve, and it might be to develop, like I say, a new platform technology. We've talked about some of those platforms. It might be to use an existing platform, like I say, to solve a specific problem. But we're always open to people coming up with ideas and doing things, I like to think, and people do. And sometimes, you know, we do additional projects if they make sense. Sometimes we get additional people to help. Like one of the things that we've often done is with maybe what I call side projects is we have undergraduates. MIT has a very strong undergraduate research program, and we have undergraduates working on aspects of them.
Brent Valentine:
[12:17] Yeah. And then how have you adapted leadership styles to inspire and motivate all the different types of people that have come through your lab? Because I'm sure now it's been hundreds of different people across generations.
Robert Langer:
[12:32] Right, right. Well, I think my leadership style, like I say, is I try. I'm not talking about I think it's maybe more inspirational. I'm also not I'm probably the opposite of a micromanager. You know, I want people to grow and have a lot of freedom, but that's kind of my leadership style, I think.
Brent Valentine:
[12:51] Yeah. And then when you have so many different projects kind of working concurrently, how do you juggle attacking all the different intricacies of each one at different times?
Robert Langer:
[13:02] Yeah. Well, I don't know that they're that different, you know, and I don't know that there's so many, but they, and they all have some pretty common themes. You know usually they involve materials and they involve medicine and you know and and there's lots of people not just me that help i mean when you have teams of people uh there's a and you're in the environment we're at i mean not even not just our lab sometimes we collaborate with others we collaborate with other people in other departments we collaborate with people in hospitals you know things like that so it's it's it's um It's pretty diverse.
Brent Valentine:
[13:40] Yeah. And then on that, you said they're not that different, each problem. And I feel like from the outside perspective, people might see like, oh, you're doing all these different things. But do you think your ability to kind of condense each individual thing to a core foundational set of principles of what allows you to simplify it and then attack the problems?
Robert Langer:
[14:04] I don't know if it's necessarily that. I mean, that probably helps. But i think the biggest thing is having really good people you know who want to attack it and have some basic principles from what we've done before and others have done and what they've done and and then they you know attack the problem as best they can
Keller Kramer:
[14:26] And with the collaboration in the lab when the problem is initially starting will there be kind of a discussion with the different subgroups of the lab about how to tackle it? Will it start kind of, if it's a materials problem, would it start with the materials team and eventually make its way to the rest of them? Or will they kind of work from the onset in a more collaborative sense to determine the direction?
Robert Langer:
[14:49] I'd say the second of those two things that you just said.
Brent Valentine:
[14:55] And then you've created different drugs or delivery mechanisms that cover a whole host of different problems. How much do you follow the real-world impact or the patient stories downstream of your innovations?
Robert Langer:
[15:12] Well, I think it depends. I mean, in the beginning, you know, first of all, it doesn't happen without companies. Sometimes we've helped start those companies. Secondly, maybe the earliest patients, they get more attention, not just from me, but from everybody. But then sometimes there's so many patients. I mean, if you take one of the companies that helped start was Moderna. So, I mean, that's probably hundreds of millions, if not billions of patients. It's not like, I mean, I know a lot of people that have had the shot, but I certainly don't follow it, for example.
Brent Valentine:
[15:52] Yeah. But when you have that large of an impact, do you ever feel a certain weight or expectation to continue innovating, continue helping people, or that type of thing?
Robert Langer:
[16:03] Well, not because of that, but I guess the way I've looked at what I do, I love being a professor. I love working with the students, and I really enjoy pushing technologies out. It's not an easy thing to do. But I suppose if what I've tried to do as I've really, as I've gotten older, maybe sort of a way of thinking about it is I try to look at, well, with whatever limited skills or whatever skills I have, what can I do that could hopefully have the greatest positive impact on the world? I mean, there's plenty of things I'm not good at, but, you know, I can, so I don't know at any particular time what I, how I'd answer that question in advance. But when I sort of see the situation, you know, then I try to do what I can.
Keller Kramer:
[16:57] And you mentioned, you know, the collaboration with business. I think that's kind of been a core aspect of your career is not only getting these technologies done on an academic sense, but actually getting them to that next phase and getting them out to people. So I guess, how do you, one, think about the relationship between business and academia? And it's always evolving. And then secondly, I think a lot of professors we've talked to might have ideas that are really strong in an academic sense, but they kind of lack that business bone. And that seems to be a hindrance for them because then they become dependent on more grant style funding. And it seems to be, I don't want to say an issue, but like we're talking about
Keller Kramer:
[17:42] the freedom that happens, it seems to limit freedom in some aspects. So I guess, how do you think about that? And what, in your experience, made you so good at kind of crossing that bridge between academia and business?
Robert Langer:
[17:55] Well, I don't know that I'm so good, but the way I looked at it is I wanted to see what we did make an impact. I really didn't know how in the beginning. um you know i it certainly seemed to me that it wouldn't happen without companies but large companies you know would initially they did give us some grants i mean took many years even before that happened but they did give us some grants but they and they were going to develop it the technologies but then they spent a year or two and gave up and one of my friends mentioned to me about starting a company ourselves. And we did. And, you know, we certainly made plenty of mistakes. I think you learn through those mistakes. I mean, it was also probably a mistake to have licenses to the large companies. But you learn each time you do it, you learn more and you get maybe a little better at it. But it's still, you know, I think it's really just, But I suppose if I had one passion or maybe one difference, it's that I really didn't want to just do the basic research. I like that, but I wanted to do something to help people.
Brent Valentine:
[19:12] Do you think the patient-centric approach and the translatability of your research is as common throughout many other labs in academia?
Robert Langer:
[19:25] Well, I think there are certainly other labs that want to help people. I think most people do. I'm not sure that any of us know how best to do it. And it's certainly a long road from basic science to medical treatment. But there's plenty of labs that contribute to that in different ways.
Brent Valentine:
[19:47] Yeah. I was just more interested on, do you think now more than ever, academia needs to be taking on that patient-centric first or the idea of how can this create real impact and that's going to allow for a more sustainable production of the research?
Robert Langer:
[20:05] Yeah, I actually don't think that, you know, I think that's one good model, but I still believe a lot in basic, you know, really fundamental research, basic curiosity driven research. But I think if you do either of those, I think you ultimately then when you make a discovery, you might think of, well, what's the next step? But I, I don't, I think it's really important to do good basic research as well as good applied research.
Keller Kramer:
[20:33] And at what point, I guess, in that discovery process, do you think that scientists should start to think about the potential next step of expanding out to business? When should someone know that a discovery is ready for the market?
Robert Langer:
[20:48] Well, I don't know that you ever know for sure, but maybe when you, if I think about it, you know, maybe when you've got an in vivo proof of principle that something is safe and effective, you have publications, you have a patent, maybe, you know, those kinds of things. yeah
Brent Valentine:
[21:04] And then do you think certain new technologies like the organ on a chip will speed up that process because we can start to see it in something like close to in vivo but at an earlier stage
Robert Langer:
[21:18] Yeah i especially i do but then it's really important that the uh um organ on a ship um really validate you know be validated for humans
Brent Valentine:
[21:33] Is that technology not validated yet by like the governing bodies to like push it along the process
Robert Langer:
[21:40] Yeah i i don't even know if it ever will be i mean but different people including ourselves have made organs on a ship but you might imagine you take it out of the human environment well how are you dealing with vascularization how are you dealing with the immune system so there's lots of things that uh you know that you don't have And so, but it's still, that doesn't mean that it can't help, but, you know, it's trying to deal with those things is important.
Keller Kramer:
[22:14] And when you think about these complicated technologies, especially delivery mechanisms like the nanotechnologies or organon chip, is there an aspect of considering how you will educate the general public on the technology? Because, you know, obviously with like the mRNA vaccine, there's a lot of pushback on, you know, whether the science was good. And a lot of people that are generally uneducated on, you know, what the technical science going on is, but they are massively opinionated. And I'd imagine like it's going to get continually more complicated as, you know, more and more of robotics or robotic adjacent technologies are brought in to medicine. How do you balance that and how do you navigate that?
Robert Langer:
[23:01] Well, I think that's a great question and I don't know that anybody's navigated that so well. I think, boy, the public, I mean, I still really don't understand how different, you know, the diversity of public opinions are so different on almost anything. I just personally have always believed, well, you try to do good science, you write it up in good journals, but some of these things are political hot potatoes. And certainly vaccines are today, which is almost shocking to me, but it is. I would expect organs on a ship to be shocking too, but at least they're not being injected. But that won't stop crazy people from talking about it and probably saying bad things about it. There's just, I don't know how you'd do it.
Robert Langer:
[24:07] Sometimes the press in the United States, they say, that they could say misleading things obviously politicians say misleading things all the time you know and it's just it's it's it's just too bad so i i don't know the answer to that i and believe me i i haven't seen anybody give me a good answer because i like to understand that myself i think it would be great if if if if we could educate people better you know there have been whole conference is dedicated to it and so forth but it goes beyond science yeah I
Brent Valentine:
[24:46] Think that's where some of the mass media that's kind of happening now media being taken into independent hands will really play a bigger role because you see other professors like Lex Friedman and them they're able to distill complex scientific ideas into easily accessible to the mass public And I would be hopeful that independent media and those types of navigations, those systems will distill these ideas into the broader sphere a lot easier without getting the politicized news article type of headline or that type of thing.
Robert Langer:
[25:24] Yeah, but there's a lot of people, I mean, I did his podcast, too, and that was great, and I think that those things are good. But there's so many not good ones, too, that also get a lot of attention.
Brent Valentine:
[25:42] Yeah. Yeah, it's a tricky one to balance.
Keller Kramer:
[25:45] Yeah do you think the responsibility as this guy is stepping a little bit more into, outside of the regular field of this conversation but does the responsibility fall on on the scientists and the people conducting the work and driving it forward to be the educators or at some point is there have to be a balance of you know the actual academic systems yeah.
Robert Langer:
[26:12] I think it's maybe everybody's responsibility. I think some people are, there's the news media who are writers, there's scientists who do the work, there's politicians who make policy. I think it's everybody, but there's not even close to uniform agreement.
Brent Valentine:
[26:32] Then kind of looking a bit more into the future, what do you think are some of the biggest problems that you want to tackle next?
Robert Langer:
[26:41] Well, we're doing a lot with the Gates Foundation trying to develop, adapt our technologies to the developing world, you know, making better medicines, making them last longer because compliance is, you know, people for not having access to taking them is an issue. We're developing vaccines that are what we call self-boosting, meaning you'd only have to take one once. We're working on better ways of nutrition. We're working on new nanotechnology approaches for targeting specific cell types in the body. And then there's all the tissue engineering work, you know, which organs on a chip, which might someday, as you pointed out, reduce or eliminate, I would probably say reduce animal and maybe human testing. And you could also do things much faster that way too to the extent that they're validated.
Keller Kramer:
[27:42] Could you explain a little bit more the self-boosting vaccine? Because I saw that the other video on it, and I just found it super interesting and a really creative way of solving what is at the end of the day of a compliance issue.
Robert Langer:
[27:55] Yeah. Well, and it may even be able to give you better immunity,
Robert Langer:
[28:00] too, and may be useful for even other things like allergies or so forth, right? Because you would just get a single shot. But basically, the idea is you take your vaccine, you put it inside a little particle. Think of it as a mini tiny eggshell, ultra tiny eggshell, and you make a bunch of these. But each eggshell has a slightly different thickness or slightly different degradation rate, even though that eggshell is made out of a polymer that is FDA approved and breaks down into water and CO2. So anyhow, you have all these different eggshells that will break it at exactly one week, two weeks, one month, two months, six months, a year. And you make a cocktail and you inject that in. And it does just what I said. You get these bursts and then it comes out at these different times. It really mimics like you go into the doctor and get in your status. Just given over one.
Keller Kramer:
[29:00] Has that been implemented in a drug that's currently on the market?
Robert Langer:
[29:04] No, no. We've been working with it in the Gates Foundation. We have published it. We've patented it. We just recently started a company to try to do what you said, but it's certainly not on the market yet.
Brent Valentine:
[29:18] Is your preference to take a technology like that and start a new company or try to license it with companies that already exist?
Robert Langer:
[29:27] I'd be happy with either, but the problem with companies that already exist, especially large companies, is it doesn't get traction. You know, they might start to think that in the beginning it will, but it doesn't usually get traction. And so over the years, I've gotten more involved in starting companies just to make sure it does get traction, doesn't get lost.
Brent Valentine:
[29:50] And then how do you convince the investor that helps you start the company that this should and will like get traction or this will work
Robert Langer:
[29:59] Well the investors are smart you know if they if they think that we have a good technology you know just like yourself you know you mentioned you thought that was neat that you think they have a good technology and and you have a good patent you know they're able to to judge you know and and then they look at the team that maybe we also have a really good ceo that makes a difference to the investors too will
Brent Valentine:
[30:24] You ever pull out of conversations with an investor when you realize they don't understand the technology at a core function. And I'm kind of thinking of this because we had a conversation with Richard Miller yesterday and he was doing aging and he's saying all this funding is going towards senescence and limiting senescence in sales. But he really believes that's fundamentally incorrect. So is there ever a point where you're just like, oh, you might be willing to give us money, but you're not the right partner for us?
Robert Langer:
[30:55] Sure. Lots of things make you not the right partner. Not just what you just said, but that's certainly one example. Other examples are investors might want a quick buck. If you're trying to do it in medicine, it's not like doing Facebook or other things. It takes a long time before you are able to get a product.
Keller Kramer:
[31:17] Do you generally feel like those timelines are respected, not necessarily just in your lab and in your work, but across people in similar fields? Do you feel like that there's a consensus that that relationship allows for the proper time? Or do people feel pressure to get something out, get a publication out before it maybe is perfectly ready or move to the next stage before it's kind of a natural progression to do so?
Robert Langer:
[31:44] Sometimes people think like that, but I think people generally, I try to encourage to think more about a really great publication, you know, and think that that people around here, I think that's what they want to have.
Brent Valentine:
[31:56] Yeah. And then on a maybe more like the investor trying to push something to market, do you think that because of your reputation or something like that, they'll listen to you or your lab saying, hey, this isn't ready yet? Or have you ever faced that difficulty of them really pushing, like something goes to market too quickly?
Robert Langer:
[32:21] Well, that would be a company, not my lab, because, you know, we don't have investors in the lab.
Robert Langer:
[32:26] Sometimes people push. I mean, they always want things as fast as they can. On the other hand, I mean, you can't cut corn. Ultimately, if it's a medical product, you have to go through the FDA and have the right safety and so forth. So, you know, it's a hard, but sure, sometimes people, some investors might want things done faster than they could be done.
Brent Valentine:
[32:50] And then do you think the FDA is ever too stringent or not stringent enough, depending on what the technology is, or maybe especially a kind of really emerging technology that they might not fully understand yet?
Robert Langer:
[33:04] Well, I think they could understand it well enough, but they use pretty good criteria on both safety, efficacy, and manufacturing to make decisions. That said, they're humans, so sometimes they happen maybe too conservative. Um you know i think you know obviously and when there's crises though like with aids and like with covid then you know they speeded things up but generally they have a very conservative attitude which probably is good you know the attitude of first do no harm
Brent Valentine:
[33:33] Yeah and then once you get past first do no harm you push the drug out it's working well at the onset and then you start to see maybe a few issues arise or people have bad reactions to it do you think there's enough of an ability to look back at maybe once a drug's been out for a couple years is this working the best way possible can we redesign this to be to have less side effects or pull it off the market and come back with something that's
Robert Langer:
[34:03] Doing something if something was bad yeah i yes i mean that that those kinds of things have certainly happened
Keller Kramer:
[34:12] Looking at kind of the FDA example, across different countries, have you seen, you know, certain relationships with regulatory bodies that are more conducive for academic research getting further? We were in, we talked to someone about organ on chips, actually, in the Netherlands, and they were talking about how, you know, on campus they have regulatory bodies that they're working with constantly. The innovation is almost driven through them. And the way that that was described to me just seemed kind of foreign. But have you seen other examples, or do you think that the way we have it in the U.S. is actually a very strong model?
Robert Langer:
[34:51] Well, I think it's a strong model, but some places, like people say, going to Australia is often faster for clinical trials, as an example. So there's different. It's not a simple thing to say.
Brent Valentine:
[35:06] Yeah.
Brent Valentine:
[35:07] I think on that is just, do you think there's ways of integrating these regulators or politicians in the design process from the earlier stages that could help push it or maybe even help the public understand it, saying, we're working on this issue. Here's the things we're tackling. And then once it's ready, people are already familiar with it.
Robert Langer:
[35:27] I think it'd be great if that would happen. But I don't know if you can get the politicians to do it.
Brent Valentine:
[35:33] Yeah, that's fair. Right.
Keller Kramer:
[35:34] And looking across, you know, for new students today, it seems like there's, you know, a million directions you could take a career, which is, I guess, constant across all timelines. But it seems like the required amount of knowledge across different disciplines seems to be increasing. And the fact that, you know, if you want to be working at the cutting edge of medical technology, you have to be, you know, incredibly competent at chemical engineering, biology, and all these different processes.
Keller Kramer:
[36:02] What advice do you have to students that are maybe feeling overwhelmed about the competencies they have to develop to really be competitive? And I guess, you know, how to find their footing and what they actually are passionate about in that process.
Robert Langer:
[36:17] Well, I think I always tell people to try to find what they're passionate about. That's really important. I don't think you have to know everything. I think you should do what you enjoy and what you feel you can make an impact on.
Brent Valentine:
[36:30] Yeah. And then do you think that mentality of, I don't need to know, but I'll go out and do it anyway, is kind of something that you've embodied and you learned along the way to just constantly be trying new things and figuring out where you fit?
Robert Langer:
[36:44] Well, you can learn as you go. I mean, you know, again, when you do research, you're trying to understand something that nobody maybe fully does understand.
Keller Kramer:
[36:55] And then as we wrap up, do you have any just broad advice to students or to anyone listening?
Robert Langer:
[37:00] Some of it goes back to what I said earlier. You know, I think you want to always find your passion. You want to dream big dreams, hopefully that can make the world a better place. Also, you know, be persistent. Don't give up on those dreams.
Brent Valentine:
[37:14] Perfect. Well, thank you so much for your time today.
Robert Langer:
[37:16] My pleasure. It's great to talk to you both. Thank you. Take care. Take care.